Buffer lectures - calculation of pH with ICE table
As it was explained on the previous page, using ICE table doesn't make much sense, as it mainly makes calculations longer, without making the result substantially better (there are many other sources of errors, which make efforts involved with using ICE table waste of time). But in some variants it is still possible, and sometimes people do try to approach the problem this way, so we will just solve a single example, the same we started our work with previously:
What is pH of the solution prepared by mixing 50 mL of 0.01 M acetic acid and 50 mL of 0.01 M sodium acetate?
Our initial concentrations (after taking dilution into account) are 0.005 M HAcetate and 0.005 M Acetate-. We will check if the acid doesn't dissociate further (or perhaps if the final pH of the solution is not dominated by the conjugate base hydrolysis):
| HAcetate | ↔ | H+ | Acetate- | |
| I | 0.005 | 0 | 0.005 | |
| C | -x | x | +x | |
| E | 0.005-x | x | 0.005+x |
when plugged into dissociation constant formula we get:
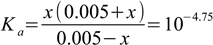 3.1
3.1which yields a simple quadratic equation. Of the two existing solutions only x=1.76576×10-5 makes sense (most of these digits are superfluous, we listed them just to not obscure the result with roundings), which means the pH is not 4.75 as calculated using Henderson-Hasselbalch equation, but 4.753. 0.003 is well below the accuracy of a typical bench pH meter. Just taking ionic strength of the solution into account changes final pH by 0.03 pH unit - ten times more, than the increase of accuracy that could be attributed to the use of ICE table.



