Percentage to molarity concentration conversion formula derivation
First of all - in many cases to be able to convert concentration you have to know molar mass of the substance and density of solution. Although every chemist should be able to convert concentrations by hand in case of necessity, it is much better to do the conversion using concentration calculator - you don't have to derive formulas, you don't have to look for density tables nor for molar masses, as these are built in into our CASC. Try it for free to see if it really saves your time.
In the most general approach to concentration conversions you always start from the definitions of the concentrations to derive conversion formula. Sometimes you may found that to proceed you need some additional information.
Let's say we have known mass percentage and we want to convert it to molarity. Molarity is defined as
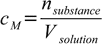 10.1
10.1where cM is a molarity, nsubstance is number of moles and Vsolution is volume. Mass percentage is defined as
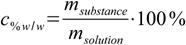 10.2
10.2What we have to do is to express both number of moles and volume of the solution using terms present in the mass percentage definition. It is obvious that both formulas will be not enough for conversion - we have to find a way to convert mass of substance to number of moles and mass of solution to its volume.
Number of moles is
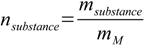 10.3
10.3where mM stands for molar mass. msubstance can be found using percentage definition:
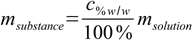 10.4
10.4so
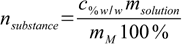 10.5
10.5What about solution volume? Once again we will need help of some additional formula. We know solution mass (msolution), we need solution volume - and these two values are tied by the solution density:
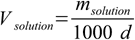 10.6
10.6Why did we use 1000 factor? Because volume is expressed in liters while density in g/mL. 1000 is a number of mL in liter.
Now we are ready to put it all together, combining 10.1, 10.5 and 10.6:
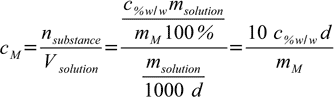 10.7
10.7or
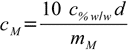 10.8
10.8Done.
At this stage it is very easy to solve this equation for mass percentage:
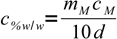 10.9
10.9and we have ready formulas for both molarity to percentage and percentage to molarity conversions.



