Concentration follies - when concentration value seems stupid
There are situations when results of concentrations calculations look either funny or suspicious. Two of them are described - and explained - below.
What is molality of 99% w/w acetic acid?
Using formula derived in the concentration conversion section we have
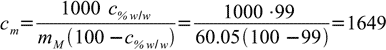 13.1
13.1(the same result can be obtained much faster using concentration calculator).
Something must be wrong. 1649 seems to be just too large.
But it isn't.
Molality is a number of moles per 1 kilogram of solvent. In 99% w/w acetic acid there are only 10g of water in every kilogram of solution. Thus we need 100 kg of solution for 1 kg of water - that gives 99 kg of acetic acid, or 1649 moles.
If the acetic acid is treated as solvent and water as dissolved substance the same solution has much more 'normal' molality of 0.5607.
What is mass-volume percentage of 80% w/w sulfuric acid?
Let's start finding conversion equation.
Mass percentage is defined as
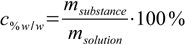 13.2
13.2Mass-volume percentage is defined as
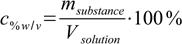 13.3
13.3msubstance is mass in g, Vsolution is volume in mL.
We know that
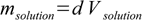 13.4
13.4putting it all together yields
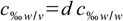 13.5
13.580 % w/w sulfuric acid has a density of 1.7323 g/mL (check our density tables or use our concentration calculator).
Thus mass-volume percentage is 1.7323×80=138.58%
Absurd? Yes. But mass-volume percentage is faulty by definition.
(this is taken from CASC help)



