pH calculator program - Base Acid Titration and Equilibria - screen shots
Once you are done with the screen shots, select example of pH calculation to learn how the dialog windows shown here can be used.
This is the main program dialog, where you define the solution. Acid and base parameters can be entered manually or selected from the database, concentration can be entered, or calculated using concentration calculator (see below). According to the picture pH of the solution made by mixing 20 mL 0.01 M malonic acid solution and 40 mL of 0.01M aniline solution is 4.56 (ionic strength ignored). Mixing known volumes of solutions is only one of the ways to prepare data for calculations.
This is the concentrations calculator - modifying any value will force other values to be recalculated. For the molar mass to be calculated it is enough to enter the chemical formula of the substance.
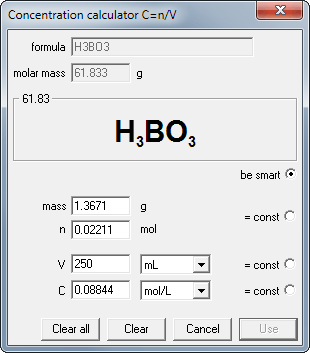
Solution window and titration view, with both titration curves - one for acid/base and one for base/acid titration. Ionic strength was calculated for every curve point.
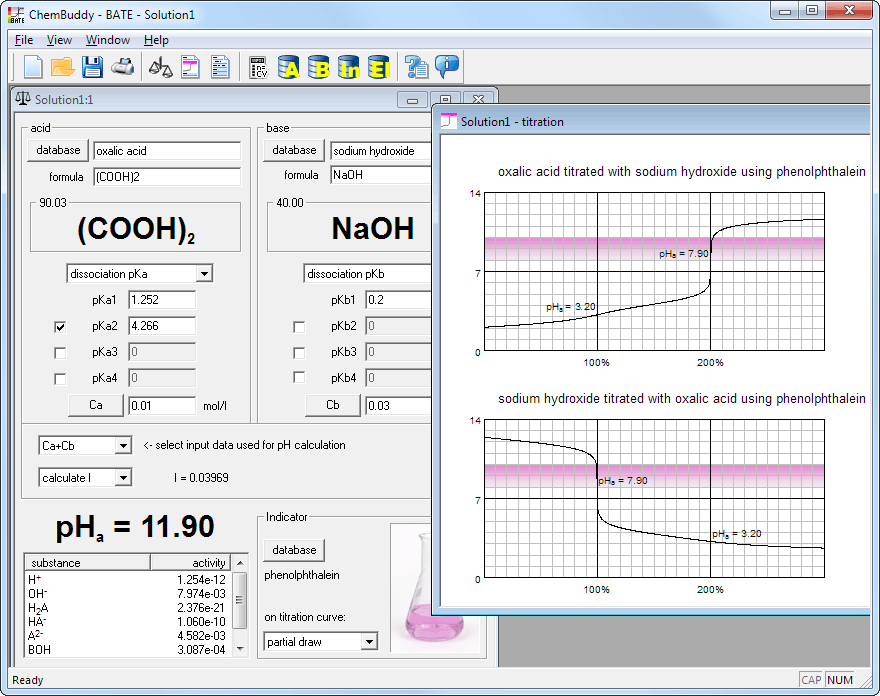
Solution window and equilibrium view, containing information about pH, activities and activity coefficients (if calculated), together with concentrations of all ions present in the solution.
Database window, where you can select substances for your solution. Program accepts constants describing acid/base strength (or weakness) as six different constant types, as every source tend to use its own convention.
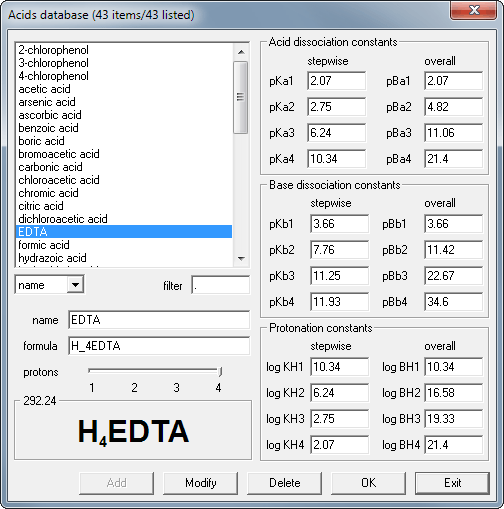
This is only a fraction of program features, feel free to browse the site to see more, or to download the free 30-day trial to try BATE by yourself. You may also download just BATE help file in Windows chm format.



