preparing chemistry quizzes and tests - concentration calculation questions
This was a standard quiz given to students to check their knowledge on the concentration calculations. As such it should contain two numerical questions. Teacher wanted to ask one question about conversion from percentage to molarity, and one about the final concentration of mixture made of two solutions. Only two versions of the questions were required this time.
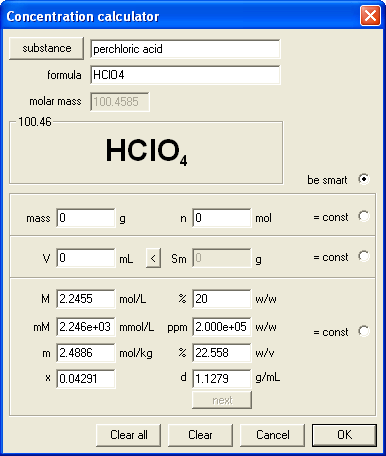
Conversion from percentage to molarity requires knowledge about solution density. As CASC (concentration calculator) contains built in density tables, that was not a problem. We decided for the simple question:
What is the molarity of ....% perchloric acid. d=.....g/mL C=.......M
Two concentrations selected were 10.0% (d=1.0597 g/mL, Cm=1.0549 M) and 20.0% (d=1.1279 g/mL, Cm=2.2455 M) - it is worth of noting that all we had to do was to select perchloric acid from the database and enter percentage concentrations, both densities and answers for answer key were immediately calculated by the program, so it turned out to be a half a minute task.
We spend much more time preparing the other question, about mixing. Not because it was difficult to use CASC, rather because it gave us too many ways of dealing with the problem. In the question were you mix two solutions and you get a third one, you may use identical concentration unit for all solutions or different units for all solutions, you may give the information about amount of the solution as volume or mass and so on. We have saved so much time during preparation of previous questions that we decided for a more relaxed and less rigorous discussion. The most ambitious version of the question proposed called for mixing of solutions with given percentage and molarity concentrations and asking for the molality of the mixture. In fact we prepared such a question and it took just a few seconds, but teacher decided against, to not scare her students. We also discussed much easier version, in which 0.1 M and 30% solutions are mixed to get known volume of 1 M solution, but again decided against, as it requires conversion of percentage to molarity, which is identical with the task assigned in the first question.
Finally, after playing with various variants of concentrations and substances, we decided for:
You have mixed 560 mL of 0.1 M ammonium nitrate solution with 413 mL of 2 M solution of the same substance. Final solution weights exactly 1000 g. What is its molality. C=......mol/kg
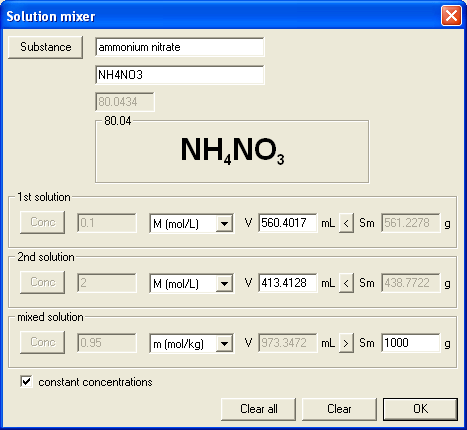
Even if at first sight it looks complicated, both number of moles of substance and mass of solvent are easy to calculate, which makes question much easier than it looks. Second version was identical, just for different substance and different molality:
You have mixed 474 mL of 0.1 M calcium nitrate solution with 424 mL of 2 M solution of the same substance. Final solution weights exactly 1000 g. What is its molality. C=......mol/kg
In both cases volumes of solutions mixed were calculated automatically for given input data (concentrations and solution mass) by the mixing calculator. In the first question correct answer is 0.95 mol/kg, in the second - 1.05 mol/kg.



