Definition of mass percentage
Mass (or weight) percentage (% w/w) is one of the most often used ways of expressing concentrations. It is defined as
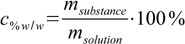 1.1
1.1Mass of solution (in case of simple solution containing only one solvent and one solute) is
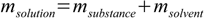 1.2
1.2so we can formulate the definition in slightly different way:
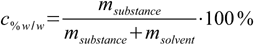 1.3
1.3Weight percentage is the only percentage concentration that is always unambiguous. Note that it is expressed in % units (as opposed to % w/v).
Unfortunately bottles labelled % often contain solution of unknown concentration. See what is wrong with percentages for detailed explanation.



