pH calculator program - Base Acid Titration and Equilibria
pH calculator is designed to help you in time consuming calculations that you may encounter in your daily practice. With acid formulas, names and dissociation constants fetched from the database, and fast numerical engine, calculation of pH of rather nasty mixtures of phosphoric acid and ammonia (five separate equilibria - three for the acid, one for ammonia, one for water) can be calculated as fast as you are able to type in concentrations - but that's not all!
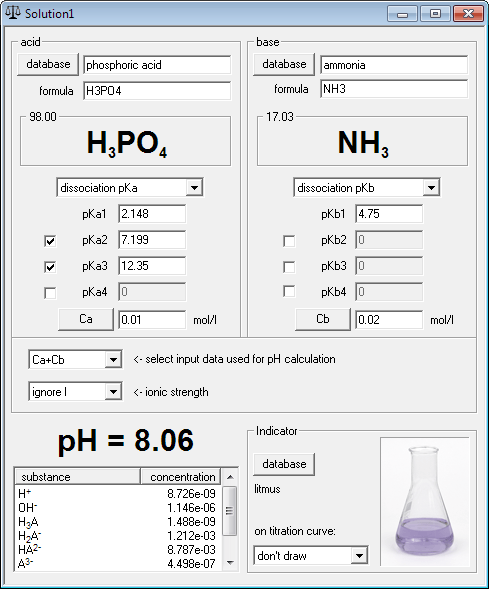
- Find - using pH calculator - exact pH of any mixture of strong/weak/polyprotic acid and base. Such mixtures include solutions of most salts and buffers.
- Calculate, ignore, or force ionic strength during pH calculation.
- Let pH calculator calculate concentrations (or activities) of all forms of dissociated acid and base present in the solution.
- Use built into pH calculator expandable acid and base database.
- Use expandable indicator database to check approximate colors of indicators in the solutions of calculated pH.
- Calculate the concentration of acids and bases using the simple concentration calculator.
- Define solution constitution in several easy ways.
- Use pH calculator to calculate the shape of virtually any acid/base titration curve.
- Let pH calculator print calculation results and titration curves for further reference.
You will find the description of algorithm used for pH calculations in the pH calculation method section. Basically it's a brute force numerical attack on a full set of equations describing acid and base dissociation equilibrium.
BATE pH calculator needs any PC class computer with properly running Windows - any version published after 1995 should do.
Note: pH calculator in BATE is limited to two component mixtures containing one acid and one base. For more flexible tool without this limitation, please check pH calculator built into our other program: Buffer Maker.
pH calculations are one of the first things taught in any general chemistry course. There are two reasons - first, pH is a very important property of the solution. Secondly, pH calculations are a very good introduction to the concept of chemical equilibrium in the solution. While in many cases pH calculations are not very difficult thanks to the approximations that can be performed. Quite often we don't know how good these approximations are, or how good is pH value calculated with their use. Lack of reasonable method of pH calculation in case of acid with similar Ka values resulted in frustration. This frustration was the mother of our first pH calculator.
The first version of the pH calculator was prepared 20 years ago on the TI-59 calculator. This was soon followed by the Sinclair Spectrum version. The first, non-commercial PC version of the pH calculator was written in the late eighties. Theoretical model used by pH calculator was improved several times, gaining speed, accuracy, and stability, but it was always based on the same numerical approach described on the site.
During these 20 years pH calculator was used for solving pH questions from the textbooks (helped find several nasty errors), for preparation of pH questions for exams, in analytical practice and as a helpful tool used in electrochemical research. Acid/base titration curves prepared with pH calculator were used in lectures in analytical chemistry course. During all these years pH calculator was a handy tool, saving us a lot of time on many occasions - and proving its accuracy and reliability.
The current version of the pH calculator is used in industrial labs, R&D labs, in pharmacological labs, by students and educators in many countries - like US, Switzerland, Spain, Mexico, Germany, Denmark, Canada or Sweden to name a few - and the family of users is growing.
Lately - together with our other calculators - pH calculator is often used to check answers to pH questions asked by students on many chemical forums.



